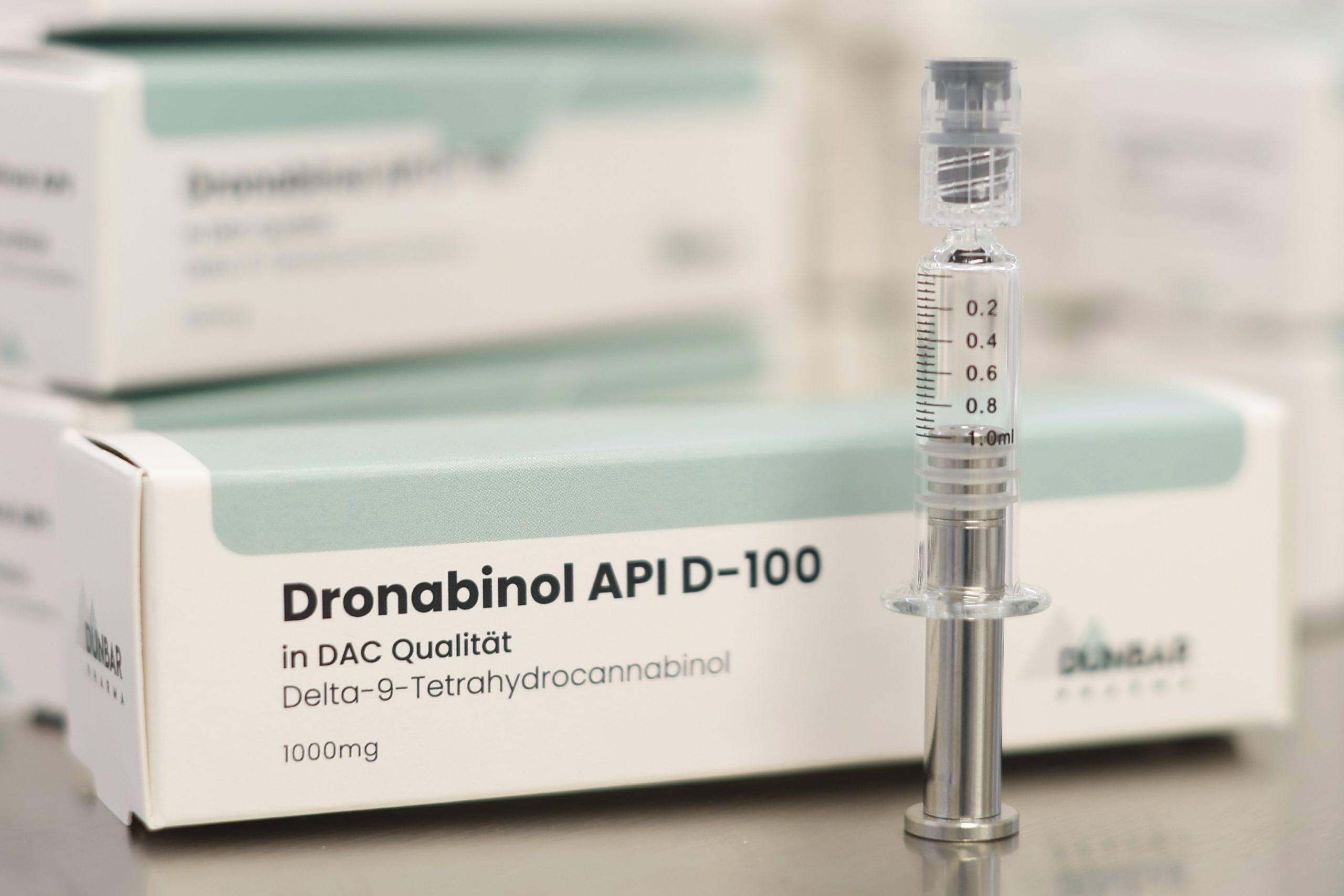
DRONABINOL Dunbar Pharma API D-100 / 1000mg
excl. 19% VAT incl. shipping costs
Product Type: Active Pharmaceutical Ingredient (API)
Manufacturer: Dunbar Pharmaceuticals Ireland
Strength: 1,000 mg Δ⁹-tetrahydrocannabinol (THC)
Form: Viscous oil
Colour: Clear to very light yellow; may show slight purple hue on the surface (per DAC/NRF specifications)
Purity: ≥ 97% Δ⁹-THC
Origin: 100% plant-derived (non-synthetic) cannabis. (Not-irradiated)
Packaging: 1 g per borosilicate glass vial
Storage: Ambient
Description:
Dunbar Dronabinol is a pharmaceutical-grade Δ⁹-tetrahydrocannabinol (THC) active substance manufactured under EU-GMP certification for use in magistral preparations.
The API fully conforms to DAC/NRF quality specifications for supply chain, certification, colour, purity, and identity, ensuring regulatory compliance and protection against pharmacy retaxation.
Quality & Compliance:
- True EU-GMP certified manufacturing and release
- Conforms to DAC/NRF specifications
- Recommended validated ID test: Farmako
- Full batch Certificate of Conformity and Certificate of Analysis provided
- High stability and batch-to-batch consistency
Intended Use:
For use in pharmacy compounding (e.g., dronabinol oils or capsules as per DAC/NRF) in accordance with applicable pharmaceutical regulations.

 Deutsch
Deutsch